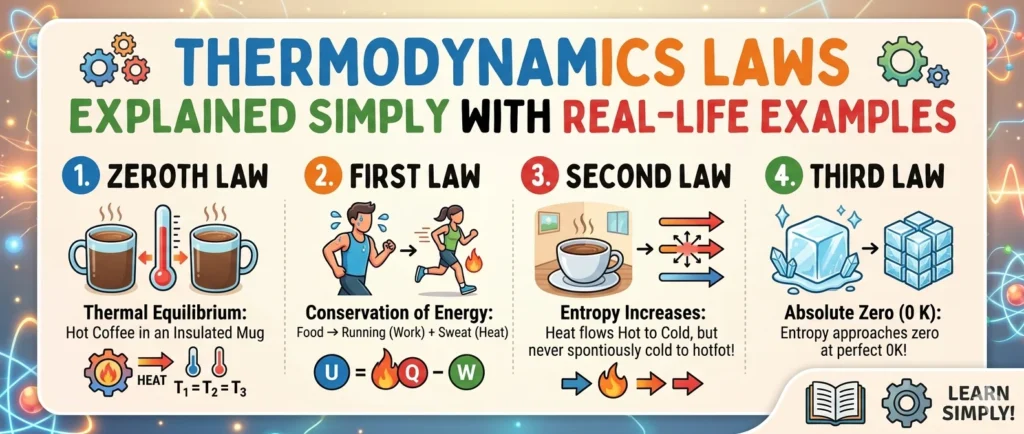
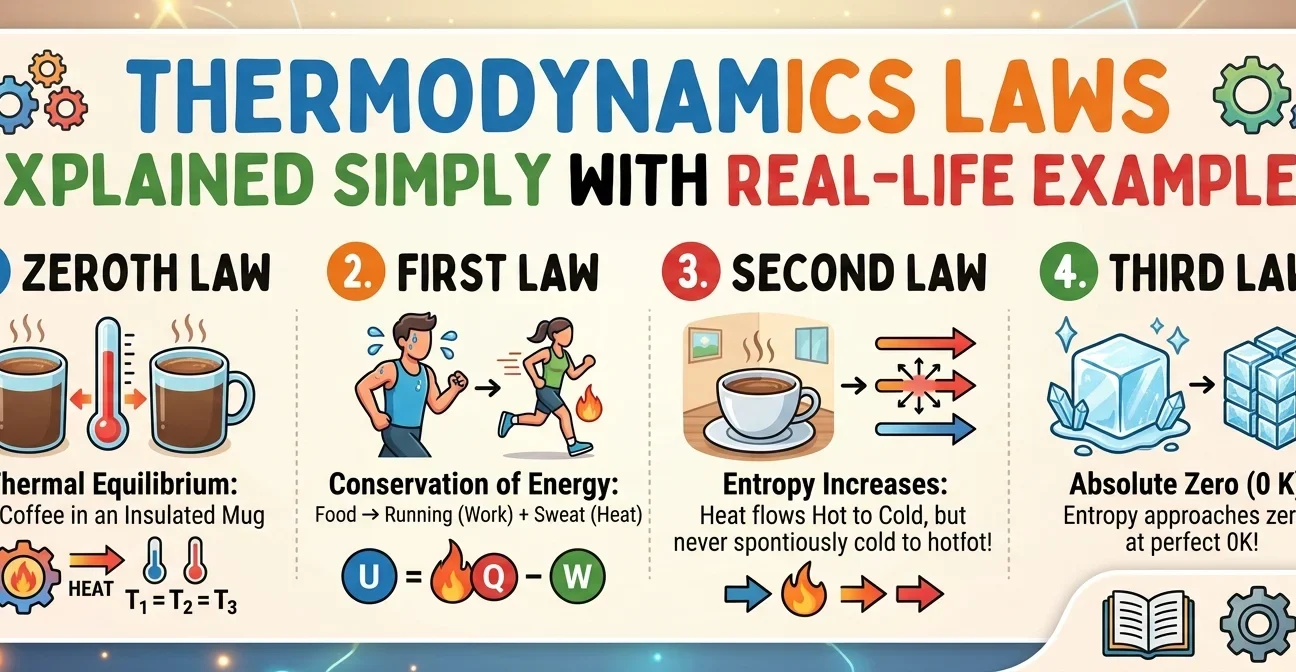
Thermodynamics Laws Explained Simply with Real-Life Examples
Have you ever wondered why your hot coffee always cools down but never heats up on its own? Or why no machine is ever 100% efficient? The answers all lie in the four laws of thermodynamics — arguably the most fundamental rules governing the physical universe. In this tutorial, we'll break each law down in plain language, connect it to things you see every day, and give you exactly what you need to understand (and remember) them forever.
First Things First: What Exactly Is Thermodynamics?
Simply put, thermodynamics is the branch of physics that studies heat, work, temperature, and energy — and how they interact. The word itself comes from Greek: therme (heat) + dynamis (power). It governs everything from the engine in your car to how stars burn, from your refrigerator to the inside of a volcano.
Thermodynamics is built on four bedrock principles, called the Zeroth, First, Second, and Third Laws. Each law captures a different truth about how energy behaves in the universe. Let's explore them one by one.
The Zeroth Law of Thermodynamics
"If A equals B, and B equals C — then A equals C."
Interestingly, this law was formulated after the First and Second Laws — but it was so fundamental that scientists had to insert it before them. Hence the name "Zeroth."
In simpler terms, if object A and object B are both at the same temperature as object C, then A and B are automatically at the same temperature too. It sounds obvious — but it's this very principle that makes thermometers possible.
🌡️ Real-Life Example: Your Thermometer
Everyday Example
When you place a thermometer under your tongue, it comes into thermal equilibrium with your body. The mercury (or digital sensor) then has the same temperature as you. Since you both reached equilibrium with the thermometer as the middleman, the reading is your temperature. That's the Zeroth Law at work — every time you check a fever.
Additionally, think about touching a metal spoon and a wooden spoon left in the same room. Both are at room temperature — meaning both are in thermal equilibrium with the room. As a result, they're in thermal equilibrium with each other, even though the metal feels colder (that's a different story about conductivity, not temperature).
The First Law of Thermodynamics
"Energy cannot be created or destroyed — only transformed."
The First Law is essentially the Law of Conservation of Energy, applied to heat and thermodynamic systems. It tells us that the total energy of an isolated system always remains constant — energy simply changes form.
ΔU = Q − W
In everyday language: if you add heat to a system, that energy goes into either increasing the system's internal energy or letting the system do work. Nothing is lost — it simply transforms. This is why "perpetual motion machines" that create energy from nothing are physically impossible.
☕ Real-Life Example 1: Boiling a Kettle
Everyday Example
When you heat water in a kettle, you're supplying electrical energy (heat, Q) to the water. Most of this energy increases the internal energy of the water molecules — raising the temperature. Eventually, some of it does work by pushing steam out of the spout. The First Law guarantees: energy in = energy stored + energy used as work.
🚗 Real-Life Example 2: A Car Engine
Everyday Example
When petrol burns in a car engine, chemical energy converts to heat. That heat then does mechanical work — pushing pistons, turning the crankshaft, and ultimately moving the car. No energy is created; it's all a series of transformations. The exhaust heat that escapes is not "wasted" in the universe's ledger — it's simply transferred elsewhere, keeping the total energy budget perfectly balanced.
🧊 Real-Life Example 3: Rubbing Your Hands Together
Everyday Example
On a cold day, you rub your palms together and feel warmth. Here, mechanical work (rubbing) converts directly into internal energy (heat) in your skin. No heat was added from outside — yet your hands got warmer, because work is a form of energy that can raise internal energy. First Law, perfectly demonstrated.
The Second Law of Thermodynamics
"Nature always moves toward disorder."
The Second Law is arguably the most profound — and the most misunderstood — law in all of physics. While the First Law says energy is conserved, the Second Law tells us something deeper: not all processes are reversible, and nature has a preferred direction.
In other words: heat flows naturally from hot to cold — never the other way on its own.
The key concept here is entropy (S) — a measure of disorder or randomness in a system. The Second Law says the universe naturally drifts toward higher entropy (greater disorder) over time. This is why things fall apart, rust, decay, and cool down — but never spontaneously organise themselves back into perfect order.
🍦 Real-Life Example 1: Ice Cream Melting
Everyday Example
Leave an ice cream cone on a table on a warm day, and it melts. Heat flows from the warmer room air into the colder ice cream. You will never see ice cream spontaneously freeze itself using the surrounding warm air. That would require heat to flow from cold to hot on its own — which the Second Law absolutely forbids.
🧊 Real-Life Example 2: The Refrigerator (Fighting the Second Law)
Everyday Example
A refrigerator does move heat from cold (inside) to hot (outside) — but notice it requires electrical energy to do so. It doesn't violate the Second Law; it uses external work to force heat in an unnatural direction. The total entropy of the system + surroundings still increases. Your fridge literally pays an entropy tax to the universe by releasing heat at the back.
💧 Real-Life Example 3: A Drop of Ink in Water
Everyday Example
Drop a single drop of blue ink into a glass of water. It gradually spreads and diffuses until the water is uniformly blue. It will never spontaneously un-mix itself back into a drop. That spreading is entropy increasing — molecules moving from an ordered (concentrated) state to a disordered (spread out) state. This is one of the most vivid demonstrations of the Second Law.
🔥 Why No Engine Is 100% Efficient
The Second Law also explains why no heat engine — a car engine, steam turbine, or power plant — can ever be perfectly efficient. Some heat must always be lost to the surroundings as "waste heat." This isn't a design flaw; it's a fundamental constraint of the universe. The best theoretical efficiency any engine can achieve is described by the Carnot efficiency, and even that is always less than 100%.
The Third Law of Thermodynamics
"Absolute zero is the unreachable finish line."
The Third Law addresses the lower limit of temperature — what happens as we cool a substance toward the coldest possible temperature, absolute zero (0 Kelvin, or −273.15°C).
In practical terms, this means two important things. First, a perfect crystal at absolute zero would have zero disorder — every atom locked in a perfectly ordered arrangement. Second, and even more striking: absolute zero can never actually be reached. You can get arbitrarily close, but crossing that finish line is physically impossible.
❄️ Real-Life Example 1: Cooling in a Laboratory
Everyday Example
Scientists can cool materials to within a billionth of a degree above absolute zero using lasers and magnetic traps. Each step of cooling, however, requires more and more effort — and the closer you get, the harder it becomes. The Third Law says you'll never quite get there. It's like running toward the horizon: no matter how fast you go, it keeps retreating.
🔬 Real-Life Example 2: Liquid Helium
Everyday Example
Liquid helium, used to cool MRI machines in hospitals, operates near 4 Kelvin — extremely close to absolute zero but still not there. At these temperatures, quantum effects dominate; helium becomes a superfluid that flows without friction. The Third Law tells us why we cannot simply cool it further to zero — entropy cannot be completely eliminated from any real system.
📊 Quick-Reference Summary Table
Now that we've covered each law in detail, here's a side-by-side comparison to help you remember them all at a glance.
| Law | Core Idea | Key Concept | Real-Life Example |
|---|---|---|---|
| Zeroth | Thermal equilibrium is transitive | Temperature | Thermometers measuring body heat |
| First | Energy is conserved; ΔU = Q − W | Internal Energy | Car engine, boiling kettle |
| Second | Entropy always increases naturally | Entropy | Ice melting, ink diffusing in water |
| Third | Entropy → 0 as T → 0 K; absolute zero unreachable | Absolute Zero | Liquid helium, MRI cooling systems |
Frequently Asked Questions
Q1. Why is it called the "Zeroth" Law and not the "Fourth"?
The concept was recognised after the First and Second Laws were already established, but scientists realised it was more foundational than both. Rather than renumber the existing laws, they inserted this one as the "Zeroth" — logically prior to all others.
Q2. Does the Second Law mean everything will eventually fall apart?
In a broad sense, yes. The Second Law implies that the universe trends toward maximum entropy — a state called "heat death," where all energy is evenly distributed and no useful work can be done. However, this is a timeline of billions of years; locally, entropy can decrease (as in living organisms) as long as even more entropy is created elsewhere.
Q3. Can we ever build a perfectly efficient engine?
No. The Second Law guarantees that some energy will always be lost as waste heat in any heat engine. The theoretical maximum efficiency (Carnot efficiency) depends on the temperatures of the hot and cold reservoirs — and it is always strictly less than 100%.
Q4. What happens to entropy in living organisms?
Living organisms maintain low internal entropy (high order) by consuming food and expelling heat — increasing the entropy of their surroundings. They don't violate the Second Law; they simply redirect it. Life is, in a beautiful sense, a local battle against entropy — one that always adds more disorder to the universe than it removes from itself.
Q5. What is the difference between heat and temperature?
Temperature measures the average kinetic energy of particles in a substance. Heat, on the other hand, is energy in transfer — it flows from higher temperature to lower temperature. The Zeroth Law defines temperature; the First Law governs heat as a form of energy; and the Second Law dictates the direction heat naturally flows.
Q6. Is thermodynamics relevant to everyday life?
Absolutely. Thermodynamics underlies your refrigerator, car engine, air conditioner, power plants, the weather, cooking, your own metabolism, and even the death of stars. Understanding these four laws is understanding why the physical world works the way it does.
Bringing It All Together
The four laws of thermodynamics together paint a remarkably complete picture of how energy works in our universe. The Zeroth Law tells us what temperature even means. The First Law assures us that energy is never lost — only transformed. The Second Law reveals that nature has a direction, always marching toward greater disorder. And the Third Law sets an ultimate, unreachable boundary at the coldest possible temperature.
Far from being dry textbook content, these laws explain the morning coffee cooling on your desk, the fuel efficiency of your car, the reason your room gets messy, and the ultimate fate of the universe. Once you see them, you see them everywhere.
Whether you're preparing for an exam or simply satisfying your curiosity, these four laws are worth knowing deeply — because they are, quite literally, the rules of the universe.